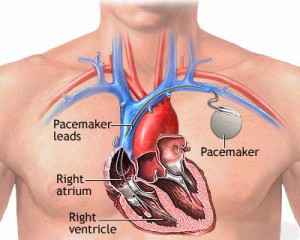
 Millions of Americans suffer from heart arrhythmia’s that are mild to moderate and some even suffer from such severe arrhythmia’s that there is a potential for death. Those at increased risk of dangerous heart arrhythmia’s or irregular heart rhythms are often treated using medical devices known as ICD (Internal Cardioverter Defibrillator, or simply defibrillator) which work to detect & change life-threatening rhythms.
Millions of Americans suffer from heart arrhythmia’s that are mild to moderate and some even suffer from such severe arrhythmia’s that there is a potential for death. Those at increased risk of dangerous heart arrhythmia’s or irregular heart rhythms are often treated using medical devices known as ICD (Internal Cardioverter Defibrillator, or simply defibrillator) which work to detect & change life-threatening rhythms.
Unfortunately, even the small, surgically implanted devices known as defibrillators, that so many arrhythmia sufferers have grown to rely upon can fail. Often times, the failure of an ICD is the direct result of manufacturing process and in many cases, these malfunctions result in a product recall. When an ICD is recalled, there is an immediate need for the patient to seek medical help from an electrophysiologist to ensure that he or she is not at risk of serious medical injury or death.
Why are defibrillators prescribed?
Defibrillators are commonly used to treat cardiac arrhythmia’s that involve a rapid heart rate (medically known as tachycardia). Defibrillators are also capable of performing as a pacemaker if needed. Once surgically implanted, the defibrillator will send regular electrical impulses to the heart to maintain a safe rhythm if necessary. Defibrillators are design to treat life threatening tachycardia; however they can also help to alleviate the symptoms of bradycardia allowing the patient to live a healthy, active lifestyle despite their heart arrhythmia. Unfortunately, when defibrillators do not operate properly, the patient is at serious risk of complications resulting mainly from the abnormal heart rhythm which is why it’s especially important for patients who have a defibrillator implanted to regularly see an electrophysiologist to ensure that the defibrillator is functioning correctly.
Defibrillator Recalls
The FDA has classified three levels of recalls which basically outline the actions that must be taken whenever a medical device is recalled. Each classification is based on the FDA findings of risk assessment and the potential threat that the defective device will cause to the patient. Defibrillator recalls that can result in severe malfunction of the device which could lead to patient death are typically Class I recalls—the most serious of the FDA medical device recall classifications.
The classifications of medical device recalls are as follows:
- Class I Recalls – issued by the FDA when there is great potential for injury or death resulting from the defective device. This is the most serious FDA recall class for medical devices such as pacemakers.
- Class II recalls – issued by the FDA when there is serious potential risk but there is only a remote possibility of death or serious injury occurring. The FDA issues this type of medical device recall when it determines that the patient may suffer reversible health consequences if the device fails but there is little reason to believe that long-term or irreversible damage will ensue.
- Class III Recalls – issued by the FDA when there is virtually no risk to the patient’s health should the device malfunction. The FDA issues this type of recall when it determines that a medical device violates the law and should be removed from the marketplace but does not pose any risk to patients.
Importance of Seeing an Electrophysiologist
Patients, who have had a defibrillator implanted, should receive regular check-ups from an electrophysiologist to ensure that the defibrillator continues to operate correctly and has not been the subject of a product recall. Complications related to defibrillators can and do occur, but in many cases they can be prevented with regular follow up care. The most common complications that arise in defibrillators involve device malfunction. This is often the result of incorrect software programming, defibrillator and/or lead failure, but may also be caused by the patient himself.
Regular health check-ups can prevent the risk of defibrillator malfunction in some cases and are recommended for anyone who has had a medical device such as a defibrillator implanted to ensure patient health and safety. Healthcare providers are typically well informed and up-to-date with the latest product recalls, especially when they pertain to their particular area of expertise. If you are a defibrillator patient, talk with your electrophysiologist about recent recalls and the steps you should take to avoid device malfunction.
Posted by: Axiom Healthcare Marketing


